Presentation of Case
Dr. Erin E. Mauney (Pediatrics): A 16-year-old girl was admitted to this hospital because of abdominal pain and bloody diarrhea.
The patient had been well until 5 weeks before this admission, when diarrhea and intermittent, diffuse, crampy abdominal pain developed. During the subsequent week, the diarrhea occurred more frequently, and the abdominal pain worsened in severity, such that it sometimes awoke the patient from sleep. Four weeks before this admission, the diarrhea began to contain dark-red blood. The patient’s mother took her to the primary care clinic of another hospital.
In the primary care clinic, the weight was 98.7 kg; the physical examination was otherwise normal. The hemoglobin level was 12.5 g per deciliter (reference range, 12.0 to 16.0). Stool cultures for salmonella, shigella, campylobacter, and Escherichia coli O157:H7 were negative, as were antigen tests for giardia, cryptosporidium, and Clostridioides difficile toxin. Empirical treatment with ciprofloxacin was started.
During the next 3 weeks, the patient completed the course of ciprofloxacin as instructed, but the abdominal pain and bloody diarrhea continued. On the day of this admission, diarrhea occurred at least four times. The patient’s parents called the primary care clinic and were instructed to bring her to the emergency department of this hospital.
On evaluation, the patient reported fatigue, malaise, weight loss, decreased appetite, and intermittent nausea and vomiting when she ate food. She also had headache and mild, diffuse joint and muscle pain. She had had no fever or sick contacts.
The patient had obesity, anxiety, and attention deficit–hyperactivity disorder (ADHD). Medications included fluoxetine and dextroamphetamine–amphetamine; there were no known drug allergies. Menarche had occurred when she was 11 years of age, and her last menstrual period had been 2 months earlier. She did not drink alcohol or use illicit drugs, and she did not vape or smoke tobacco. The patient lived in a rural area of New England with her mother, father, and sister, as well as her pet dog, cat, and lizard. She attended a technical high school with an emphasis on agriculture, and she had frequent interactions with marine and farm animals through school. Her family history included colorectal cancer and amyotrophic lateral sclerosis in her maternal grandfather, diverticulosis in her maternal grandmother and mother, psoriasis in her maternal grandmother and uncle, and celiac disease in her paternal cousin. Her father had hyperlipidemia, and her mother had had one miscarriage, which had occurred in the first trimester of pregnancy.
On examination, the temporal temperature was 36.4°C, the heart rate 112 beats per minute, and the blood pressure 94/68 mm Hg. The weight was 89.8 kg, and the body-mass index (the weight in kilograms divided by the square of the height in meters) was 32.7. The patient appeared pale, tired, and uncomfortable because of pain. The mucous membranes were dry. The abdomen was nondistended, and there was mild tenderness in both lower quadrants. There was no ulceration or rash.Table 1.  Laboratory Data.
Laboratory Data.
The white-cell count was 10,690 per microliter (reference range in children 12 to 17 years of age, 4500 to 10,700), the platelet count 188,000 per microliter, and the hemoglobin level 9.8 g per deciliter. The blood level of C-reactive protein was 248.0 mg per liter (reference range, 0.0 to 8.0), and the erythrocyte sedimentation rate was 71 mm per hour (reference range, 0 to 20). The blood levels of electrolytes and glucose were normal, as were the results of kidney-function tests. Other laboratory test results are shown in Table 1. Testing for severe acute respiratory syndrome coronavirus 2 was negative. Intravenous fluids, acetaminophen, and ondansetron were administered, and the patient was admitted to this hospital.
On hospital day 2, the hemoglobin level was 8.2 g per deciliter, and one unit of packed red cells was transfused. Esophagogastroduodenoscopy (EGD) and colonoscopy were performed.Figure 1.  Images Obtained during Esophagogastroduodenoscopy and Colonoscopy.
Images Obtained during Esophagogastroduodenoscopy and Colonoscopy.
Dr. Maureen M. Leonard: On EGD, two cratered esophageal ulcers were located 30 cm from the incisors (Figure 1A). The proximal, middle, and distal portions of the esophagus were normal. Diffuse mild inflammation — characterized by erythema and friability — was found in the cardia, gastric fundus, and gastric antrum (Figure 1B). The duodenum appeared normal. Biopsy specimens were obtained throughout the procedure for histologic evaluation.
When the patient was being positioned on the procedure table for the colonoscopy, a large volume of bright-red blood was noted to be draining from the rectum. Perianal and digital rectal examinations did not reveal a source of bleeding. Emergency radiography of the abdomen did not reveal free air, and the colonoscopy was started. On colonoscopy, the rectum and proximal sigmoid colon appeared normal (Figure 1C). However, as the colonoscope was advanced farther into the sigmoid colon, multiple nonobstructing, large, friable, dark-violet lesions were identified and biopsied (Figure 1D, 1E, and 1F). Because of narrowing and severe friability, the area was not traversed with the endoscope, and the procedure was aborted.
Dr. Mauney: The patient was transferred to the pediatric intensive care unit of this hospital. During the subsequent 2 days, an additional two units of packed red cells were transfused. Treatment with pantoprazole was started.Figure 2.  Initial Imaging Studies.
Initial Imaging Studies.
Dr. Sjirk J. Westra: Computed tomography (CT) of the abdomen and chest was performed (Figure 2A through 2D). There was irregular thickening of the large bowel wall and adjacent fat stranding, with sparing of the distal sigmoid colon and rectum. The kidneys had multifocal patchy areas of hypoattenuation.
Dr. Mauney: On hospital day 5, tachycardia and tachypnea developed. The hemoglobin level was 8.8 g per deciliter, and the platelet count was 67,000 per microliter. The prothrombin time was 17.3 seconds (reference range, 11.5 to 14.5), the prothrombin-time international normalized ratio 1.4 (reference range, 0.9 to 1.1), the activated partial-thromboplastin time 50.0 seconds (reference range, 22.0 to 36.0), and the blood d-dimer level greater than 10,000 ng per milliliter (reference range, 0 to 500). Other laboratory test results are shown in Table 1.
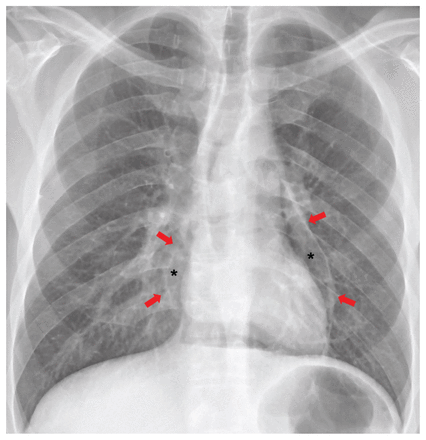
Dr. Westra: Thoracic CT angiography revealed a central filling defect in the distal right pulmonary artery, a finding consistent with pulmonary embolism (Figure 2E). There was also a large right pleural effusion.
Dr. Mauney: A diagnostic test was performed.
Differential Diagnosis
Dr. Stacy A. Kahn: The process of generating a differential diagnosis is iterative. The framework shown below — gather, analyze, review and revise, and create a differential diagnosis — is a simple approach that reminds us that the differential diagnosis often evolves and should be reviewed and revised as we gather new information and analyze the data.
Gather
This patient is a 16-year-old girl with a history of obesity, anxiety, and ADHD who presented with a 5-week history of worsening abdominal pain and diarrhea that progressed to bloody diarrhea. Stool studies were negative, and she had no response to treatment with ciprofloxacin. Three weeks after starting the course of antibiotics, she presented with anorexia, weight loss, nausea, vomiting, fatigue, malaise, headache, mild arthralgias, and myalgias. On examination, she was afebrile and appeared tired, uncomfortable, and pale, with dry mucous membranes. She had lower abdominal tenderness but no rash or jaundice.
Analyze
We can start the diagnostic process by ruling out diagnoses that do not fit with this patient’s clinical picture. Polyps and polyposis syndromes, Meckel’s diverticulum, and vascular malformations can be ruled out because these conditions are not painful or associated with weight loss or systemic symptoms. Intussusception causes abdominal pain but is unlikely in this patient because it is not associated with joint pain, weight loss, or systemic symptoms. Henoch–Schönlein purpura causes abdominal pain and can be associated with joint pain and weight loss, but the hallmark feature is skin involvement, which was not present in this patient. We can also rule out Helicobacter pylori infection and peptic-ulcer disease. These conditions can cause abdominal pain, weight loss, nausea, and vomiting, but without diarrhea or chronic symptoms. In addition, patients with H. pylori infection or peptic-ulcer disease would typically present with reflux symptoms and acute hematemesis or melena. Myalgias and arthralgias are not part of the clinical presentation.
Review
This patient’s initial laboratory evaluation revealed a borderline elevated white-cell count, anemia, hypoalbuminemia, a normal platelet count, mildly elevated aminotransferase levels, and a normal bilirubin level. The C-reactive protein level and erythrocyte sedimentation rate were markedly elevated.
Create a Differential Diagnosis
Patients with inflammatory bowel disease (IBD) — including Crohn’s disease and ulcerative colitis — typically present with bloody diarrhea, abdominal pain, anorexia, weight loss, nausea, vomiting, fatigue, malaise, and arthralgias, with no response to antibiotics. These findings are similar to those observed in this patient. Her borderline elevated white-cell count, elevated erythrocyte sedimentation rate, anemia, and hypoalbuminemia could be consistent with IBD. However, her C-reactive protein level was higher than would be expected with this condition, and she did not have thrombocytosis, which is typical of a flare of IBD. Nevertheless, IBD is still high on the list of possible diagnoses, and further evaluation that includes upper endoscopy and colonoscopy is warranted.
Other chronic conditions, particularly rheumatologic diseases, should be included in the differential diagnosis, given the elevated C-reactive protein level and erythrocyte sedimentation rate, as well as the arthralgias, fatigue, and malaise. The predominance of gastrointestinal symptoms and the absence of arthritis, rash, and fever would be somewhat atypical for a rheumatologic diagnosis. Additional laboratory testing that includes tests for antinuclear antibodies and complement levels will be important.
Although the stool studies were negative, infection should still be considered, particularly zoonotic infection because of the patient’s exposure to animals at school and at home. However, she had no fever, rash, or respiratory or neurologic symptoms.
Like IBD, cancer can be associated with many of the signs and symptoms that were observed in this patient, including anorexia, weight loss, fatigue, and a markedly elevated C-reactive protein level. However, she had no fever, night sweats, or lymphadenopathy.
Revise
After the patient was admitted to the hospital, colonoscopy was performed and revealed findings that were highly suggestive of severe colonic ischemia.1 The differential diagnosis should be revised to include colonic ischemia, a relatively rare diagnosis in children and young adults. Potential causes of colonic ischemia include infections such as coronavirus disease 2019, multisystem inflammatory syndrome in children, tuberculosis, and sepsis. Because the patient had been exposed to agricultural settings, we should consider farm-related toxins, such as insecticides, rodenticides, snake venom, and spider toxins. Drug-induced colonic ischemia is rarely reported in children and is associated with the use of nonsteroidal antiinflammatory drugs, oral contraceptives, antipsychotic medications, illicit drugs, immunosuppressants, and vasoconstrictors, none of which this patient had taken. Cardiovascular disease and end-stage kidney disease can cause colonic ischemia but are not consistent with this patient’s history.
CT imaging was also performed. In addition to findings in the bowel, there was patchy hypoattenuation in the kidneys, which suggests the possibility of ischemia or septic embolism.
On hospital day 5, the patient’s clinical status suddenly worsened, with new tachypnea and tachycardia. The white-cell count increased, and she had worsening anemia, thrombocytopenia, and elevation of the aminotransferase levels. The activated partial-thromboplastin time was prolonged and the d-dimer level was markedly elevated, but the international normalized ratio was relatively normal. Additional imaging studies revealed findings consistent with pulmonary embolism. At this point, several organs — including the lungs, the kidneys, the colon, and possibly the liver — had evidence of thrombotic injury or ischemia. Disseminated intravascular coagulation (DIC) is a consideration, but the relatively normal international normalized ratio is not consistent with this diagnosis. Also, with florid DIC, the severity of illness would be even greater. It is notable that the patient was in a hypercoagulable state in the context of a prolonged activated partial-thromboplastin time. The differential diagnosis should be revised to include hematologic conditions such as bleeding disorders, macrophage activation syndrome, and hemophagocytic lymphohistiocytosis, as well as rheumatologic conditions such as systemic lupus erythematosus, protein C or protein S deficiency, factor V Leiden, systemic juvenile idiopathic arthritis (Still’s disease), vasculitis, and antiphospholipid syndrome (APS).
Overall, this patient had evidence of acute ischemia involving multiple organ systems, without neurologic, dermatologic, or cardiac involvement. She had no evidence of sepsis, infection, or cancer. The family history included autoimmune disease (celiac disease and psoriasis) and first-trimester spontaneous abortion. She was in a hypercoagulable state with thrombosis but did not have DIC. This unusual presentation is so severe and dramatic that it is catastrophic. Taking this all into account, I think that this patient’s diagnosis is catastrophic APS (CAPS).
Dr. Stacy A. Kahn’s Diagnosis
Catastrophic antiphospholipid syndrome.
Diagnostic Testing
Figure 3.  Colon-Biopsy Specimens.
Colon-Biopsy Specimens.
Dr. Sarah B. Mueller: Histologic examination of biopsy specimens of the rectum, sigmoid colon, and distal transverse colon revealed normal colonic mucosa with occasional dilated crypts (Figure 3A). A biopsy specimen of the transverse colon from the area adjacent to the mass showed marked mucosal edema, hemorrhage, distorted colonic crypts, and multiple fibrin thrombi (Figure 3B). No classic changes associated with ischemic injury were identified, and immunohistochemical stains for cytomegalovirus and herpes simplex virus types 1 and 2 were negative. Taken together, these findings were not consistent with IBD and were instead suggestive of a thrombotic disorder.
An initial blood specimen was sent to the coagulation laboratory on hospital day 2. The prothrombin time was within the normal range (14.4 seconds), and the activated partial-thromboplastin time was prolonged (53.6 seconds). Testing for factor Xa was negative, which indicated the absence of heparin, low-molecular-weight heparin, heparin analogues, and factor Xa inhibitors. Testing for lupus anticoagulant was positive and did not show correction in a 1:1 mix of the patient’s plasma with normal plasma, which indicated the presence of an inhibitor (e.g., a lupus anticoagulant), rather than a factor deficiency. The confirmatory hexagonal phase phospholipid neutralization assay was positive. Taken together, these results are considered to be positive for a lupus anticoagulant. However, it is important to note that the patient’s C-reactive protein level was highly elevated on admission. C-reactive protein can bind to the phospholipids that are present in the reagents of the lupus anticoagulant assay and the hexagonal phase assay, and the hexagonal phase assay is known to have false positive results when the C-reactive protein level is similar to the level observed in this patient.2-5 Testing for anticardiolipin IgG and IgM, anti–β2-glycoprotein I IgG and IgM, antiprothrombin IgG, antiphosphatidylserine IgG and IgM, and anti–phosphatidylserine–prothrombin IgG and IgM was negative.
A specimen obtained for a full hypercoagulability panel was sent to the coagulation laboratory on hospital day 5. The prothrombin time and activated partial-thromboplastin time were prolonged, and both were corrected in a 1:1 mix. These findings are consistent with factor deficiencies. The levels of factors V, VII, XI, and XII were low; the levels of natural anticoagulants protein C and antithrombin III were low; the level of protein S was low-normal; and the levels of factors II, VIII, IX, and X were normal. These results are suggestive of evolving liver dysfunction. Although the results of liver-function tests were only mildly abnormal on admission, a specimen obtained later in the day on hospital day 5 showed clinically significant elevation in the levels of aspartate aminotransferase and alanine aminotransferase (Table 1). Testing for activated protein C resistance, which is primarily used to screen for factor V Leiden, was normal, as was genetic testing for prothrombin G20210A. Hereditary protein C and antithrombin III deficiencies were ruled out on subsequent testing.
The diagnostic criteria for APS that have been proposed by the International Society on Thrombosis and Haemostasis require laboratory identification of a lupus anticoagulant, anticardiolipin antibody, or anti–β2-glycoprotein I antibody in two blood specimens, with testing performed 12 or more weeks apart and within 5 years after an imaging-confirmed thrombotic event or pregnancy complication.6 Therefore, the diagnosis at this stage in the patient’s course was presumptive, pending confirmation of a lupus anticoagulant or relevant antibody after at least 12 weeks.
Pathological Diagnosis
Antiphospholipid syndrome.
Hospital Course
Dr. Nora AlFakhri (Pediatrics): Treatment with high-dose methylprednisolone, intravenous immune globulin (IVIG), rituximab, and eculizumab was started. On hospital day 9, the aminotransferase levels increased, coagulopathy worsened, and new encephalopathy and lactic acidosis developed; these changes were suggestive of acute liver failure. Additional imaging studies were obtained.Figure 4.  Additional Imaging Studies.
Additional Imaging Studies.
Dr. Westra: Doppler ultrasonography of the liver revealed a lack of flow in the main portal vein and its branches, a finding indicative of occlusive portal vein thrombosis (Figure 4A). CT angiography of the abdomen and pelvis confirmed the presence of portal vein thrombosis and revealed additional thrombi in the superior mesenteric vein, splenic vein, inferior mesenteric vein, and inferior vena cava (Figure 4B and 4C). There was extensive ischemic infarction in the periphery of the liver and the left kidney.
Dr. AlFakhri: An infusion of heparin was started. A transjugular intrahepatic portosystemic shunt (TIPS) was placed, and thrombectomy of the portal vein and catheter-assisted thrombolysis of the superior and inferior mesenteric veins were performed. However, on hospital day 10, the encephalopathy worsened, and there was massive lower gastrointestinal bleeding. Red cells were transfused, the heparin infusion was stopped, and protamine was administered. Flexible sigmoidoscopy did not identify a source of bleeding. Additional imaging studies were obtained.
Dr. Westra: CT angiography revealed residual thrombi in the splanchnic venous bed that led to revision of the TIPS and imaging-guided thrombectomy of the superior mesenteric vein and splenic vein (Figure 4D, 4E, and 4F). CT of the head revealed extensive thrombosis in the intracranial venous sinuses and left internal jugular vein, findings that were confirmed by means of magnetic resonance angiography.
Discussion of Management
Dr. Jonathan S. Hausmann: CAPS is thought to result from the binding of antiphospholipid antibodies to cell surfaces, which activates endothelial cells, monocytes, and platelets and leads to inflammation, complement activation, and thrombosis. The formation of thrombi in patients with antiphospholipid antibodies is thought to be a multihit process.7 The presence of antiphospholipid antibodies in the blood is the first event, but the antibodies typically do not cause disease until another event occurs. In a registry of children with CAPS, the most common second event was infection (61%). Cancer (17%), surgery (7%), and lupus flares (4%) were other common triggers for CAPS in children.8 Children are more likely than adults to have primary APS (68% vs. 60%), rather than APS that is secondary to a connective-tissue disease. Children are also more likely than adults to have CAPS as the first manifestation of APS (87% vs. 45%) and to have infection as a trigger for the catastrophic event (61% vs. 27%).8
On the basis of the patient’s history, she may have had several risk factors for the development of CAPS. Her mother was noted to have an early pregnancy loss; recurrent early pregnancy losses are characteristic of obstetrical APS. Some studies show that persons may have a genetic predisposition to APS, especially in the presence of some HLA alleles.7 In addition, the patient’s initial diarrheal illness, which was potentially acquired through animal exposure, may have been the trigger for her thrombotic event. However, she did not appear to have connective-tissue disease associated with her CAPS; testing for antinuclear antibodies was negative, as was testing for anti–double-stranded DNA, anti-Smith, anti–U1-ribonucleoprotein, anti-Ro, and anti-La antibodies.
The management of diseases on the APS spectrum needs to be highly individualized and targeted to the patient’s risk factors, antibody profile, and disease manifestations. Factors that increase the risk of thromboembolism include the presence of lupus, prothrombotic mutations, and traditional cardiovascular risk factors.9 Among the antiphospholipid antibodies, lupus anticoagulant is associated with the highest risk of clotting. However, the presence of a greater number of antiphospholipid antibodies at high titers confers a greater risk of thrombosis.10
Finally, thrombotic disease manifestations are important in guiding therapy. For patients with antiphospholipid antibodies and no history of clotting, anticoagulation for primary thromboprophylaxis is generally not recommended, because the annual incidence of acute thrombosis is low.11 For patients with antiphospholipid antibodies and lupus, the administration of low-dose aspirin may be beneficial, even in the absence of a history of thrombosis.9
For patients with antiphospholipid antibodies and a history of unprovoked thrombosis (e.g., patients with APS), long-term thromboprophylaxis is recommended. The standard of care for these patients is warfarin, which is administered to maintain an international normalized ratio of 2.0 to 3.0. Direct oral anticoagulants do not appear to provide any benefits and incur excess risk.12 Low-dose aspirin may be added in patients with arterial thrombosis.
For patients with more severe presentations — such as this patient, who had CAPS with thrombosis in multiple organs — treatment is more aggressive and involves targeting multiple steps within the CAPS cascade. Treatment for CAPS first addresses any precipitating factors that may have triggered the episode, if they are known (e.g., antibiotics taken for an infection). Additional treatment involves inactivating or eliminating the antibodies (plasma exchange, rituximab, or IVIG), reducing inflammation (glucocorticoids or IVIG), preventing complement activation (heparin or eculizumab), and decreasing thrombosis (heparin).
Although data from randomized trials to guide treatment for CAPS in children are lacking, treatment is generally composed of triple therapy, which includes glucocorticoids, anticoagulation, and plasma exchange or IVIG. In a registry of patients with CAPS, all the children who were treated with triple therapy survived the catastrophic event, whereas 26% of the patients in the overall pediatric cohort died.13 More recently, rituximab (an anti-CD20 monoclonal antibody) and eculizumab (which inhibits complement activation) have been used in children with CAPS, with reports of good efficacy.14
In this patient, once the diagnosis of CAPS was made, treatment with methylprednisolone, IVIG, rituximab, and eculizumab was started. Treatment with anticoagulation appears to have been delayed, perhaps because of concerns about bleeding. A heparin infusion was started after additional thrombi in portal and mesenteric vessels were identified.
Follow-up
Dr. AlFakhri: Serial CT of the head and CT angiography of the chest, abdomen, and pelvis were performed, and there was no evidence of worsening of the thrombosis or infarction. However, on hospital day 26, abdominal pain developed with enteral feeding, and another episode of massive lower gastrointestinal bleeding occurred.
Dr. Westra: CT angiography of the abdomen and pelvis revealed intraperitoneal free air, a finding suggestive of perforation due to colonic ischemia.
Dr. AlFakhri: A subtotal colectomy with creation of a Hartmann’s pouch and end ileostomy was performed because of the colonic perforation. After the surgery, treatment with low-molecular-weight heparin was started as a bridge to warfarin therapy. During the subsequent weeks, enteral feeding was restarted, and no further symptoms of colonic ischemia occurred.
Dr. Mueller: Six weeks after hospital admission, testing for lupus anticoagulant was negative, as was testing for anticardiolipin IgG and IgM and for anti–β2-glycoprotein I IgG and IgM. It is possible that the initial lupus anticoagulant assay had been falsely positive in the context of the elevated C-reactive protein level. Alternatively, the repeat lupus anticoagulant test could have been negative because of the aggressive and early treatment with methylprednisolone, IVIG, rituximab, and eculizumab. The patient continued to receive treatment for CAPS, including methylprednisolone that was transitioned to prednisone. Nine weeks after hospital admission, the patient was discharged to a rehabilitation facility.
Five months after hospital admission, repeat testing for lupus anticoagulant was initially positive but showed correction in a 1:1 mix, a pattern that is expected in the context of factor deficiencies due to warfarin therapy. The confirmatory hexagonal phase assay was negative. Testing for anticardiolipin IgG and IgM and for anti–β2-glycoprotein I IgG and IgM remained negative. Repeat imaging studies showed resolution of the sinus thrombosis and pulmonary embolism, as well as a patent TIPS and splanchnic circulation. The patient continues to receive low-dose prednisone, rituximab, and warfarin. There are plans for ostomy reversal.
Final Diagnosis
Catastrophic antiphospholipid syndrome.
Source: NEJM


 Laboratory Data.
Laboratory Data. Images Obtained during Esophagogastroduodenoscopy and Colonoscopy.
Images Obtained during Esophagogastroduodenoscopy and Colonoscopy. Initial Imaging Studies.
Initial Imaging Studies. Colon-Biopsy Specimens.
Colon-Biopsy Specimens. Additional Imaging Studies.
Additional Imaging Studies.